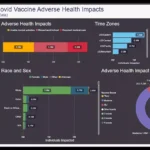
Pfizer, under the leadership of CEO Albert Bourla, has faced severe criticism for disseminating misleading information about the efficacy and safety of its COVID-19 vaccine. Dr. Bourla’s claims about the benefits of vaccinating children aged five to 11 were specifically singled out as misleading by the UK's pharmaceutical watchdog, which accused the company of failing to provide comprehensive safety data alongside its efficacy claims.
Regulatory Reprimands and Legal Actions
The UK's Prescription Medicines Code of Practice Authority (PMCPA) has reprimanded Pfizer multiple times, highlighting a recurrent pattern of unethical behavior in promoting its vaccine. The complaints ranged from promotional tweets lacking safety information to broader allegations of misrepresenting vaccine data. These actions have culminated in legal challenges, forcing Pfizer to release data and information it had withheld.

The Consequences of Corporate Misconduct
Pfizer’s actions have sparked widespread concern, calling into question the transparency and ethics of its operations. The repeated breaches of regulatory standards have not only damaged the company's reputation but also cast a shadow over the pharmaceutical industry's credibility. Critics argue that the penalties imposed on Pfizer are insufficient given the potential risks to public health and the extent of the company's misconduct.
Detailed Examination of Pfizer's Violations
The PMCPA scrutinized Pfizer's promotional activities, particularly focusing on tweets made by senior executives that implied the COVID-19 vaccine's high efficacy without adequately addressing its safety profile. These communications, deemed by the regulatory body as a proactive promotion of an unlicensed medicine, led to Pfizer's admission of guilt in misleading the public.
The specific case that drew attention involved tweets claiming high efficacy rates of the vaccine, which were posted without accompanying safety data, violating the established pharmaceutical advertising standards. This incident was not isolated but part of a series of actions by Pfizer that the PMCPA found to contravene the ethical and professional standards expected within the pharmaceutical industry.
The Ruling and Its Implications
The PMCPA’s ruling against Pfizer highlighted a significant breach of trust and raised questions about the integrity of vaccine promotion practices. The authority's decision emphasized the importance of presenting a balanced view of both the benefits and risks associated with vaccines, critiquing Pfizer for its one-sided promotional strategy.
The ruling not only mandated Pfizer to rectify its communication practices but also served as a stark reminder to the pharmaceutical industry about the paramount importance of transparency and accuracy in public health communications.
This case has reiterated the necessity for pharmaceutical companies to adhere to rigorous ethical standards in their information dissemination, especially when it concerns products with widespread public health implications.
Pfizer's behavior, characterized by misleading public statements and a lack of transparency, has significantly tarnished its reputation and underscored the need for stringent regulatory oversight in the pharmaceutical industry.

Carl Riedel is an experienced writer and Open Source Intelligence (OSINT) specialist, known for insightful articles that illuminate underreported issues. Passionate about free speech, he expertly transforms public data into compelling narratives, influencing public discourse.